Lumbar spine disc prosthesis
The visco-elastic disc prosthesis (VTDR) is a new visco-elastic disc prosthesis that is used in cases of disc degeneration. The unique design of this latest generation prosthesis has the following advantages: - Restoration of normal mobility and strength of the affected motion segment. - True-to-life 3D imitation of the human intervertebral disc. - Normal cushioning effect as with the natural intervertebral disc. - Full load-bearing capacity of the spine. - Restoration of the natural function of vertebral joints, muscles, ligaments and tendons.

What happens after the operation?
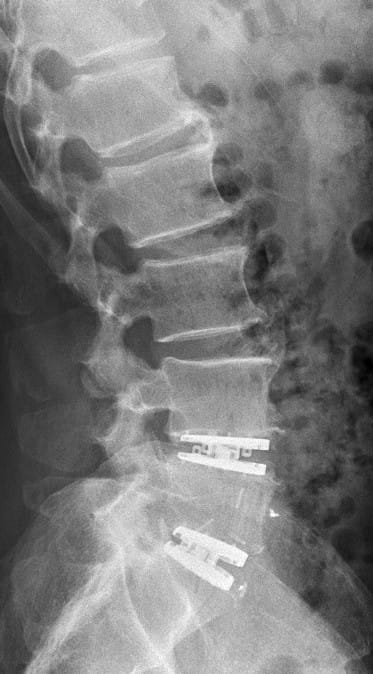
You will remain in hospital for a few days after the operation. This is usually around 3–4 days after an operation on the cervical spine and around 6–7 days after an operation on the lumbar spine. As the prosthesis is normally immediately stable in terms of movement and pressure, you will be allowed to get up just one day after the procedure. In contrast to fusion surgery, you may sit during this time, but you should avoid leaning forward under stress, hyperextension, lifting heavy objects and abrupt twisting of the spine. However, “natural” movements are permitted. This is followed by special follow-up treatment (physiotherapy, mobilisation, muscle building, etc.). After approximately 3 months, the prosthesis is usually firmly integrated into the bone. This is checked during a follow-up examination using X-rays. Mandatory follow-up examinations and documentation of findings in the SWISS Spine Register are carried out at regular intervals, guaranteeing you the greatest possible safety and care with this surgical method.
References
Berg, Js., Anthony, T., Bruski, R., et al. (2009) Total Disc Replacement Compared to Lumbar Fusion: a Randomised Level I trial With 24 Year Follow Up, Eurosurgery Spine, Volume 18, Issue 10, pages 515-524; Publisher: Springer Berlin Heidelberg New York
Zigler, Seiderman R., Isaacs R.E. et al. (2011) Results of the Prospective, Randomised, Multicentre Food and Drug Administration Investigational Device Exemption Study of the ProDisc-L Total Disc Replacement versus Circumferential Fusion for the Treatment of 1-Level Degenerative Disc Disease, Spine, Volume 13, Issue 5, Pages 501-511
Aesculap Implant Systems, Volume 18, Issue 01, Page 02 – 86), Springer Med Verlag Berlin Heidelberg New York
Fernström, U. (1966) Arthroplasty with intercorporal endoprosthesis in herniated disc and in painful disc. Acta Chir Scand Suppl., 357:154-159, Acta Chirurgica Scandinavica
Single-Level Lumbar Disc/Spondylolisthesis; Back Pain With Total Disc Arthroplasty Utilising the ProDisc-LPSPN, Volume 31, Number 10, Barnes, Page 1659–68, Lippincott Willkins & Wilkins Inc., Philadelphia
Zigler, H. E. d’Altri, H. E., Smoot, B. R., et al. (2013) Results of the Prospective, Randomised, Multicentre Food and Drug Administration Investigational Device Exemption Study of the ProDisc-C Total Disc Replacement versus Anterior Discectomy and Fusion for the Treatment of 1-Level Symptomatic Cervical Disc Disease, Spine Journal, Volume 13, Issue 10, Pages 1370-1378
